
2026 NMPA Guidelines Revisions Plan
chinameddevice.com
May 18, 2026, 9:47 a.m.
The guidelines are introduced by NMPA Center of Medical Device Evaluation (CMDE). It is not legally binding but highly recommended by regulatory authorities. The revision plan will not only impact the new product registration but also renewals and modifications. It involves type testing, clinical, registration review guidelines, technical guidelines, e.g., Real World Study design, and documentation guidelines, e.g., Indication for Use (IFU) writing. NMPA issues new or updated guidelines throughout the year. Given their importance in understanding what the NMPA reviewers are looking for, it is essential that manufacturers keep up to date with the guidelines that are latest to their specific products. One source to get updates on these guidelines is through our complimentary monthly news roundup that you can opt in from our website www.ChinaMedDevice.com
Share on

Stroke thrombectomy beyond 24 hours improves functional outcome but carries increased cerebral haemorrhage and mortality risks
neuronewsinternational.com
May 18, 2026, 9:46 a.m.
Mechanical thrombectomy can lead to an improvement in 90-day functional outcome compared to standard medical care in anterior-circulation large vessel occlusion (LVO) stroke patients presenting 24–72 hours after symptom onset, as per the findings of the LATE-MT randomised controlled trial (RCT). However, within the trial, thrombectomy was also associated with increased rates of mortality, symptomatic intracranial haemorrhage (ICH) and other clinically relevant adverse events.
Share on

Study in Chinese Neurosurgical Journal Shows Minimally Invasive Techniques
bioengineer.org
May 18, 2026, 9:45 a.m.
A pioneering study led by Dr. Youxiang Li at Beijing Tiantan Hospital, Capital Medical University, together with Dr. Wei Feng from Songyuan Jilin Oilfield Hospital, offers a robust analysis of treatment outcomes for BTAs in the modern era defined by flow diverter innovations. Published in the March 2026 issue of the Chinese Neurosurgical Journal, this important research fills a notable void in neurosurgical literature by systematically evaluating the efficacy and safety of minimally invasive endovascular treatments in a sizable cohort despite the aneurysms’ rarity.
Share on

IA Thrombolysis After Thrombectomy: More Uncertainty
www.medscape.com
May 18, 2026, 9:45 a.m.
The strategy of administering intra-arterial (IA) thrombolysis immediately after thrombectomy in patients with acute ischemic stroke has come under renewed scrutiny after results from the TECNO trial showed no clear improvement in reperfusion rates with this approach.The idea of giving a dose of IA thrombolysis immediately after thrombectomy in patients with acute ischemic stroke has become more uncertain, with the results of a new trial showing no clear benefit on reperfusion rates using this approach.These results are in contrast to the previous CHOICE-2 trial, first presented earlier this year at the International Stroke Conference, and now published in JAMA. CHOICE-2 showed positive results in terms of good functional outcomes with an IA thrombolysis strategy, although this was dampened by an increased mortality rate.
Share on

Stent-assisted coiling and flow diverters effectively treat rare basilar artery cases
www.news-medical.net
May 18, 2026, 9:44 a.m.
Patients in the study underwent a range of minimally invasive procedures, including simple coiling, stent-assisted coiling, and flow diverters, devices designed to redirect blood flow away from the aneurysm. Stent-assisted coiling was the most common treatment, used in just over half of cases, while flow diverters were employed in nearly 30 %, particularly for more complex or larger aneurysms.The results were largely positive. At follow-up imaging, about 72 % of patients achieved complete aneurysm occlusion, meaning the aneurysm was fully sealed off, while nearly 19 % had near-complete occlusion. Clinically, about 89 % of patients had favorable outcomes, defined as minimal or no disability.
Share on

In situ impedance analysis device for clot characterization in large vessel occlusion acute ischemic stroke
jnis.bmj.com
May 17, 2026, 3:13 p.m.
Large vessel occlusion (LVO) stroke is associated with a high risk of permanent disability. There are no reliable tools providing periprocedural information on clot characteristics, such as composition and length, that may impact first pass effect, which has been shown to improve functional outcomes. In the first study of a novel technology, we evaluated whether the Clotild Smart Guidewire System (Clotild), an impedance-based device, can provide in situ information on clot characteristics during a mechanical thrombectomy procedure.
Share on

TECNO: intra-arterial tenecteplase safe but does not improve reperfusion after incomplete thrombectomy
neuronewsinternational.com
May 11, 2026, 6:55 a.m.
According to the TECNO researchers, these results highlight the need for further refinement of patient selection and treatment protocols in this setting to tackle the “common and clinically important challenge” of incomplete reperfusion after a thrombectomy.TECNO was a multicentre, randomised, open-label, blinded-endpoint trial designed to test whether a 3mg dose of direct intra-arterial tenecteplase in addition to best medical treatment improves reperfusion in patients with incomplete post-thrombectomy reperfusion. The trial was coordinated by researchers from Switzerland and conducted across multiple high-volume stroke centres in Europe.
Share on

CASES: stenting during thrombectomy fails to fully establish non-inferiority versus deferred treatment in tandem lesions
neuronewsinternational.com
May 11, 2026, 6:54 a.m.
The CASES trial investigated the optimal management of acute ischaemic stroke patients with coexisting intracranial large vessel occlusion (LVO) and carotid artery stenosis ≥50% or occlusion, otherwise known as tandem lesions. Investigators compared immediate CAS versus deferred management with the aim of demonstrating the non-inferiority of immediate CAS performed during thrombectomy.CASES is an open-label, multicentre, international randomised controlled trial (RCT) that recruited patients from 26 centres across the Netherlands and Belgium. Eligible patients had ischaemic stroke due to a tandem lesion and were suitable candidates for thrombectomy. Patients were randomised to CAS at the time of thrombectomy, or to deferred treatment, which included carotid endarterectomy (CEA), delayed CAS or best medical management.
Share on

Debate heats up following contrasting trial results on brain cooling during stroke thrombectomy
neuronewsinternational.com
May 11, 2026, 6:51 a.m.
“CHILL-ART and FOCUS suggest that intra-arterial cooling remains a promising neuroprotective adjunct to thrombectomy,” ESO guideline board co-chair Diana Aguiar de Sousa (University of Lisbon, Lisbon, Portugal) told NeuroNews. “While CHILL-ART provides an encouraging efficacy signal, FOCUS is more reassuring on safety than on functional benefit. Taken together, these studies support further investigation—ideally through larger, standardised, preferably international trials designed to clarify protocol, patient selection and the consistency of the treatment effect.”
Share on

Intravenous Tenecteplase Administered Before Endovascular Therapy Shows Promise for Ischemic Stroke Patients Within 4.5 to 24 Hours
bioengineer.org
May 11, 2026, 6:47 a.m.
Tenecteplase, a genetically engineered variant of tissue plasminogen activator, has been considered a promising thrombolytic agent due to its longer half-life and ease of administration via single bolus injection. Its theoretical advantage in dissolving clots and facilitating more effective EVT has been supported by some preliminary trials. Yet, the current investigation challenges the routine use of tenecteplase as a bridging agent within the extended therapeutic window.
Share on

Efficacy and safety of a new mechanical balloon-based flow diverter in the treatment of intracranial aneurysms
jnis.bmj.com
May 8, 2026, 10:40 p.m.
The mechanical balloon-based FD showed a remarkable occlusion rate alongside minimal ischemic and hemorrhagic adverse events compared with existing FDs. This innovative mechanical balloon-based design may be an important direction for future FD design.
Share on
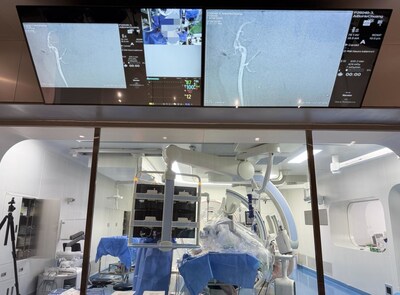
PANVIS STAR Vascular Interventional Robotic System Redefines ...
finance.yahoo.com
May 8, 2026, 10:19 p.m.
The PANVIS STAR Vascular Interventional Robotic System represents a significant advancement in treating ischemic stroke through mechanical thrombectomy. With stroke affecting millions globally and ischemic stroke comprising over 65% of cases, the demand for effective interventional solutions is critical. This robotic system addresses key challenges including limited patient access, operator fatigue, and steep learning curves by enabling remote-controlled procedures with enhanced precision. Demonstrated through successful animal model testing, PANVIS STAR successfully executed complete thrombectomy workflows including femoral access, vessel navigation, and clot retrieval. The system's innovative four-device co-manipulation capability, sub-millimeter precision, and haptic feedback enable operators to achieve superior outcomes while reducing the technical burden on healthcare professionals, potentially expanding access to this life-saving treatment globally.
Share on

Consensus statement seeks to standardise evaluation of robotic ...
neuronewsinternational.com
May 8, 2026, 10:19 p.m.
An international consensus statement has been published in the Journal of the American Heart Association establishing the first standardized framework for designing, testing, and evaluating robotic systems used in stroke thrombectomy. Led by King's College London, the statement addresses a critical gap in the field, as previous studies employed inconsistent methodologies, tested different tasks, and measured disparate outcomes, preventing meaningful comparisons. The international expert panel, comprising specialists in interventional neuroradiology, robotics, data science, health economics, policy, and patient advocacy, developed comprehensive guidelines to ensure patient safety while facilitating remote stroke treatment procedures. This standardization is expected to accelerate the advancement of robotic thrombectomy technology and expand access to emergency stroke care.
Share on
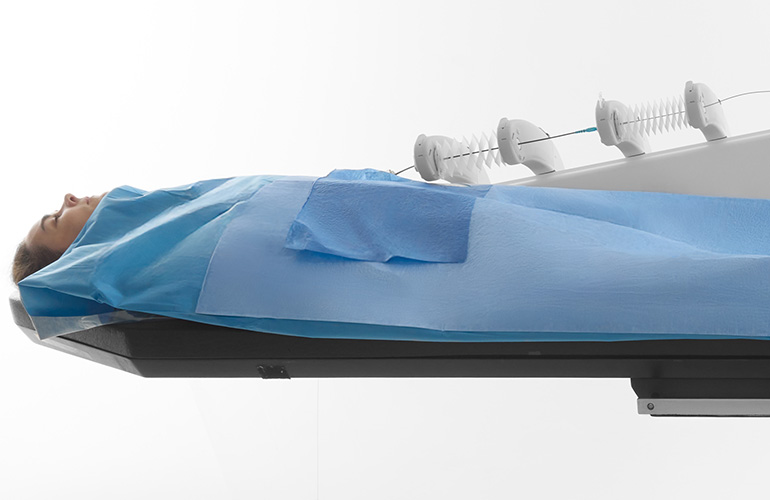
Sentante's breakthrough telesurgery system could redefine stroke care
www.medicaldesignandoutsourcing.com
May 8, 2026, 10:19 p.m.
Sentante has developed an innovative telesurgery system that could transform acute ischemic stroke treatment globally. The Sentante Stroke System, which recently received FDA Breakthrough Device designation, enables remote thrombectomies through a haptic robotic bedside unit controlled by endovascular specialists from distant locations. A landmark transatlantic demonstration in November 2025 showcased a Florida-based neurosurgeon successfully performing a procedure on a cadaver in Scotland. This technology addresses critical gaps in stroke care access, particularly for remote hospitals lacking local thrombectomy capabilities or timely transfer options. By combining digital innovation with robotic precision, Sentante's platform aims to enhance patient outcomes, improve efficiency, and ensure equitable access to life-saving interventions while addressing workforce sustainability challenges in endovascular medicine.
Share on
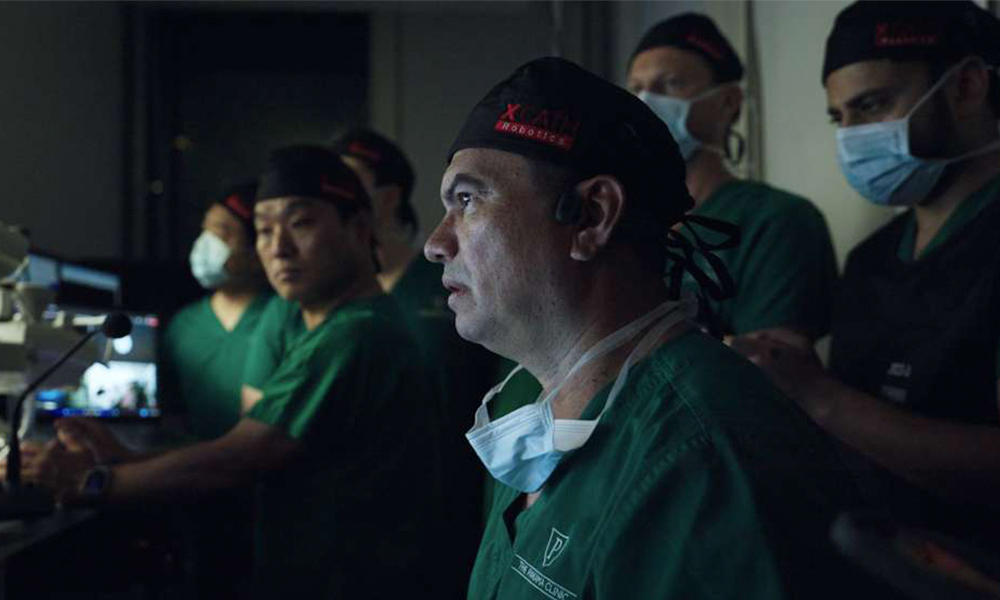
UAE-funded XCath completes world's first-in-human telerobotic stroke ...
www.crescententerprises.com
May 8, 2026, 10:19 p.m.
UAE-funded XCath, a neuroendovascular surgical robotics company partially owned by Crescent Enterprises, has successfully completed the world's first-in-human telerobotic stroke surgery. The landmark procedure involved a remote robotic mechanical thrombectomy performed using the XCath Iris Endovascular Robotic System, conducted by Dr. Vitor Mendes Pereira. This groundbreaking achievement represents a significant advancement in minimally invasive surgical technology and remote medical intervention capabilities, demonstrating the potential for telerobotic systems to enhance stroke treatment outcomes while expanding access to specialized surgical expertise across geographical distances.
Share on

Liberty - Microbot Medical
www.microbotmedical.com
May 8, 2026, 10:18 p.m.
Microbot Medical's LIBERTY® Endovascular Robotic System represents a transformative advancement in peripheral vascular intervention. This innovative single-use, remotely operated platform enables physicians to perform precise endovascular procedures from a seated position, significantly enhancing control and ergonomics. LIBERTY® eliminates the need for substantial capital investment while delivering a remarkable 92% reduction in radiation exposure compared to traditional manual techniques. The system integrates seamlessly into existing workflows with setup completed in under five minutes and maintains universal compatibility with standard guidewires and catheters. By combining compact design with intuitive remote operation, LIBERTY® modernizes vascular access and navigation, offering clinicians improved efficiency and peace of mind without the complexity associated with conventional capital equipment solutions.
Share on

Blood return system dramatically reduces blood loss during pulmonary thrombectomy
www.news-medical.net
April 27, 2026, 7:39 a.m.
The first comparative analysis from the ENGULF (A Safety and Feasibility Single-Arm Study of a Novel Catheter Thrombectomy Device For the Treatment of Pulmonary Embolism) trial shows that using blood return during continuous aspiration thrombectomy dramatically reduces blood loss by 97%, while maintaining hemodynamic efficacy and improving procedural efficiency. Researchers presented the late-breaking data today at the Society for Cardiovascular Angiography & Interventions (SCAI) 2026 Scientific Sessions & Canadian Association of Interventional Cardiology/Association Canadienne de cardiologie d'intervention (CAIC-ACCI) Summit in Montreal.
Share on

Albumin Plus Endovascular Therapy Boosts Stroke Outcomes
bioengineer.org
April 27, 2026, 7:38 a.m.
In a groundbreaking advancement for stroke therapy, a recent randomized clinical trial published in Nature Communications brings to light the synergistic effects of albumin administration combined with endovascular therapy for treating acute ischemic stroke patients. Acute ischemic stroke, a leading cause of disability and mortality worldwide, results from an abrupt obstruction of cerebral blood flow, often eliciting irreversible brain damage if not rapidly managed. While mechanical thrombectomy through endovascular techniques has revolutionized treatment by physically removing the occlusion, therapeutic adjuncts to enhance outcomes remain a critical unmet need. This study, led by Liu, Dong, Zhang, and colleagues, pioneers the exploration of albumin—a naturally abundant plasma protein—as a potential amplifier of neuroprotection and vascular restoration when paired with endovascular intervention.
Share on

Penumbra unveils latest iteration of thrombectomy system for venous thromboembolism
vascularnews.com
April 26, 2026, 6:55 p.m.
“Engineered for the rapid removal of pulmonary and venous thrombus, the latest Flash 3.0 algorithm is designed for enhanced clot detection capabilities with increased sensitivity to thrombus and blood,” Penumbra reported in a press release. The latest iteration includes enlarged tubing coupled with an automated backflush feature tailored for large thrombus burdens and to reduce friction from aspirated thrombus.The STORM-PE randomised controlled trial, recently published in Circulation, found that the use of Penumbra’s CAVT technology with anticoagulation achieved superior reduction in right heart strain compared to anticoagulation therapy alone in patients with acute intermediate-high risk pulmonary embolism (PE). This marks the first level-1 evidence demonstrating superior results for mechanical thrombectomy over traditional anticoagulation for the treatment of pulmonary embolism (PE).
Share on
E2 lands $80M series C for next-gen thrombectomy treatment
www.fiercebiotech.com
April 8, 2026, 8:54 p.m.
California-based medtech E2 (Endovascular Engineering) has raised $80 million in a series C funding round as it looks to commercialize its pulmonary embolism treatment. The Hēlo Thrombectomy Platform received FDA clearance late last year for use in peripheral veins to treat pulmonary embolism without the need for surgical intervention. The innovation behind the tech is its dual-action mechanism, which combines aspiration with mechanical clot disruption and is delivered through a small-profile catheter.
Share on