Active Post-Market Surveillance of Innovative Devices for Valvular ...
clinicaltrials.gov
June 7, 2026, 9:51 a.m.
This prospective, multicenter, observational cohort study aims to establish a post-market registry framework to evaluate the real-world clinical applicability, long-term safety, and effectiveness of innovative heart valve devices in China.
Share on
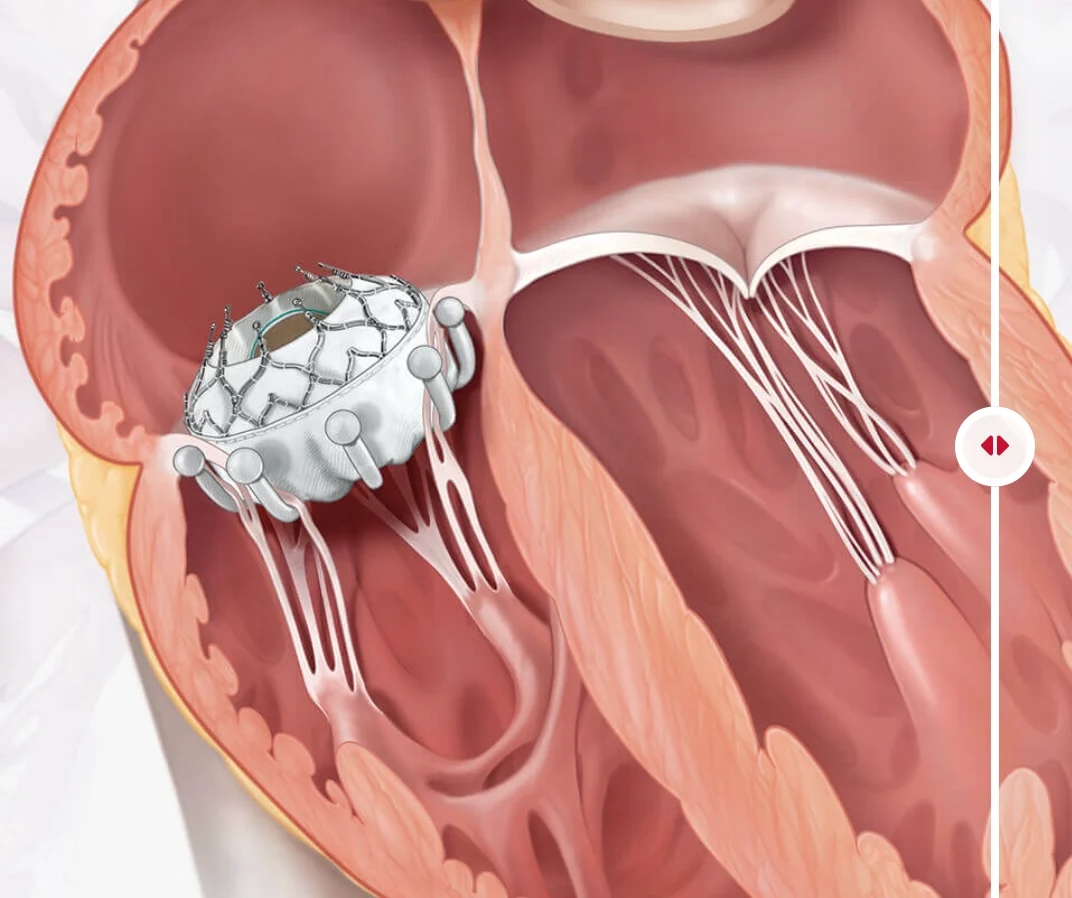
Innovations in transcatheter tricuspid valve interventions
www.annalscts.com
June 7, 2026, 9:49 a.m.
Transcatheter tricuspid valve interventions represent a rapidly evolving field shaped by early clinical experiences and iterative innovation. The formative period of development involved exploratory approaches derived from mitral valve techniques, which yielded important lessons from both successes and setbacks. Early obstacles, such as technical complexity and imaging limitations encountered with direct annuloplasty approaches like the Cardioband system, redirected research efforts and deepened understanding of tricuspid anatomy and physiology. These foundational experiences have informed current therapeutic strategies and established parameters for future innovations. The article examines what has failed, what has proven effective, and emerging approaches in transcatheter tricuspid valve intervention, providing valuable insights for the cardiothoracic surgery community navigating this dynamic treatment landscape.
Share on
Robotic-assisted Percutaneous Coronary Intervention
www.frontiersin.org
June 7, 2026, 3:13 a.m.
Robotic-assisted percutaneous coronary intervention represents an emerging advancement in coronary revascularization techniques. This article examines the current evidence and future potential of robotic systems in PCI procedures, comparing them to conventional manual techniques. While manual PCI remains the standard approach for treating coronary artery disease across acute and chronic presentations, robotic-assisted methods offer potential advantages including reduced operator radiation exposure and improved procedural precision. The review discusses applications in various clinical scenarios, from chronic coronary syndromes to acute myocardial infarctions, while emphasizing the importance of individualized patient management strategies and the need for continued investigation into when robotic assistance may provide clinical benefits.
Share on

2026 NMPA Guidelines Revisions Plan
chinameddevice.com
May 18, 2026, 9:47 a.m.
The guidelines are introduced by NMPA Center of Medical Device Evaluation (CMDE). It is not legally binding but highly recommended by regulatory authorities. The revision plan will not only impact the new product registration but also renewals and modifications. It involves type testing, clinical, registration review guidelines, technical guidelines, e.g., Real World Study design, and documentation guidelines, e.g., Indication for Use (IFU) writing. NMPA issues new or updated guidelines throughout the year. Given their importance in understanding what the NMPA reviewers are looking for, it is essential that manufacturers keep up to date with the guidelines that are latest to their specific products. One source to get updates on these guidelines is through our complimentary monthly news roundup that you can opt in from our website www.ChinaMedDevice.com
Share on

TruLeaf Medical, a fully-owned subsidiary of Allmed Solutions successfully Completed the Second Stage of valve implantation in two Additional Patients
www.prnewswire.com
May 11, 2026, 6:57 a.m.
TruLeaf Medical, Ltd, a fully-owned subsidiary of Allmed Solutions (TASE: ALMD), a clinical-stage medical device company specializing in transcatheter structural heart innovations, is pleased to announce the successful completion of the full two-stage percutaneous heart valve implantation procedure in two additional high-risk patients suffering from severe, refractory heart failure due to leaky tricuspid valve (TR). The first stage of the docking device implantation was performed 3 months ago. In recent days, the second stage of fully percutaneous transcatheter heart valve implantation within the TruLeaf RoseDocdocking station, was successfully performed at a leading medical center in India under a compassionate-use framework. This milestone achievement, conducted in collaboration with a prominent international medical device partner, further validates the clinical utility of TruLeaf Medical RoseDoc TTVR system for high-risk, no-other-option patients suffering from severe refractory heart failure due to severe TR.
Share on
Surgical Mitral Valve Replacement Shows Long-Term Survival Advantages
www.dicardiology.com
May 9, 2026, 9:44 a.m.
A new multicenter study published in The Annals of Thoracic Surgery finds that reoperative surgical mitral valve replacement (rSMVR) is associated with significantly better long-term survival compared to transcatheter mitral valve-in-valve (mViV) procedures in patients with failing bioprosthetic mitral valves. While both approaches demonstrated similar safety and procedural success at 30 days, key differences emerged over time, particularly beyond the first year.

ACC.26 Structural Intervention Trials Offer Viable Options
www.acc.org
May 9, 2026, 9:40 a.m.
Recent clinical trials presented at ACC.26 demonstrate promising advances in structural cardiac interventions. The PROTECT H2H trial evaluated embolic protection devices during transcatheter aortic valve implantation, comparing the Emboliner device with the standard Sentinel device across 522 patients. Despite periprocedural stroke occurring in only 2-4% of TAVI cases, it remains a significant concern. The trial assessed 30-day major adverse cardiovascular and cerebrovascular events as its primary endpoint. Additional late-breaking trials included PRO-TAVI, which compared routine percutaneous coronary intervention strategies with deferred approaches in TAVI patients, and SURViV, examining redo surgery versus transcatheter valve-in-valve procedures for mitral bioprosthetic dysfunction. These trials collectively provide clinicians with viable alternative options for managing complex structural cardiac conditions.
Share on
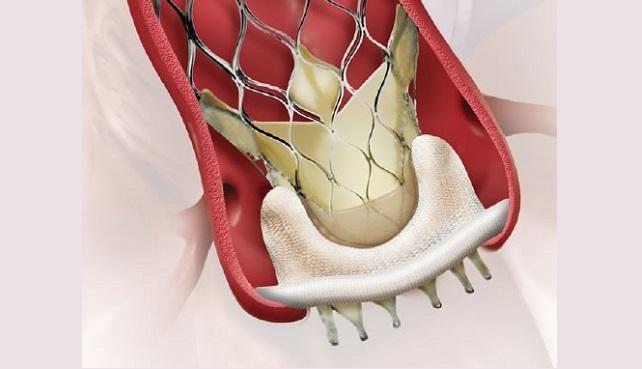
Redo surgical mitral valve replacement shows long-term survival advantage over transcatheter valve-in-valve
www.eurekalert.org
May 9, 2026, 9:39 a.m.
A multicenter study published in The Annals of Thoracic Surgery demonstrates that reoperative surgical mitral valve replacement provides superior long-term survival outcomes compared to transcatheter mitral valve-in-valve procedures for patients with failing bioprosthetic mitral valves. Analyzing 229 patients from two high-volume centers between 2004 and 2023, researchers found comparable safety profiles and procedural success within 30 days. However, significant divergence emerged beyond one year, with surgical patients achieving substantially lower five-year mortality rates of 20.3% versus 40.9% for transcatheter procedures, alongside superior valve durability and lower gradients. While transcatheter approaches offered shorter hospital stays and fewer early complications, the study emphasizes that surgical intervention delivers greater long-term benefits in appropriately selected patients. These findings support multidisciplinary heart team decision-making, balancing short-term advantages of transcatheter procedures against the enduring survival benefits of surgical replacement, particularly for low-risk patients with extended life expectancy.
Share on

Long-Term Survival Advantage of Reoperative Surgical Mitral Valve Replacement Over Transcatheter Mitral Valve-in-Valve
www.annalsthoracicsurgery.org
May 9, 2026, 9:36 a.m.
Although mViV demonstrated comparable 30-day MVARC outcomes, rSMVR was associated with superior long-term survival and hemodynamic durability among patients deemed appropriate surgical candidates by a multidisciplinary heart team. Treatment decisions should balance short-term risk with long-term outcomes.
Novel minimally invasive treatment demonstrates significant sustained improvement for heart valve patients with limited options
www.eurekalert.org
May 9, 2026, 9:35 a.m.
Researchers have demonstrated significant sustained clinical benefits from a novel minimally invasive treatment for mitral regurgitation patients who lack conventional surgical options. The AltaValve Early Feasibility Study, presented at the 2026 SCAI Scientific Sessions in Montreal, shows one-year improvement in patients treated with atrial fixation Transcatheter Mitral Valve Replacement. Mitral regurgitation, affecting over 175 million people globally, occurs when the mitral valve fails to close properly, allowing blood to leak backward. This emerging technique offers hope for elderly patients and those with anatomical constraints unsuitable for traditional interventions by placing a prosthetic valve above the diseased valve, reducing left ventricular outflow tract obstruction risks.
Share on
Transcatheter Mitral Valve-in-Valve Superior to Redo Surgery in ...
medicaldialogues.in
May 8, 2026, 11:16 p.m.
Recent clinical evidence demonstrates that transcatheter mitral valve-in-valve procedures offer superior outcomes compared to traditional redo surgery in patients with failed mitral bioprostheses. This minimally invasive approach provides significant advantages in terms of reduced procedural risk, shorter recovery times, and improved patient safety profiles. The transcatheter technique represents an important advancement in interventional cardiology, offering a viable alternative for high-risk surgical candidates requiring mitral valve replacement. These findings suggest a paradigm shift in treatment strategies for managing bioprosthetic valve failure, potentially expanding treatment options and improving clinical outcomes for affected patients.
Share on

Minimally invasive treatment demonstrates sustained improvement for heart valve patients with limited options
medicalxpress.com
May 8, 2026, 5:44 p.m.
Findings from the AltaValve Early Feasibility Study suggest that patients with a leaky mitral valve have sustained clinical benefits one year following atrial fixation Transcatheter Mitral Valve Replacement (TMVR). Researchers presented the late-breaking data at the Society for Cardiovascular Angiography & Interventions (SCAI) 2026 Scientific Sessions & Canadian Association of Interventional Cardiology/Association Canadienne de cardiologie d'intervention (CAIC-ACCI) Summit in Montreal.
Share on
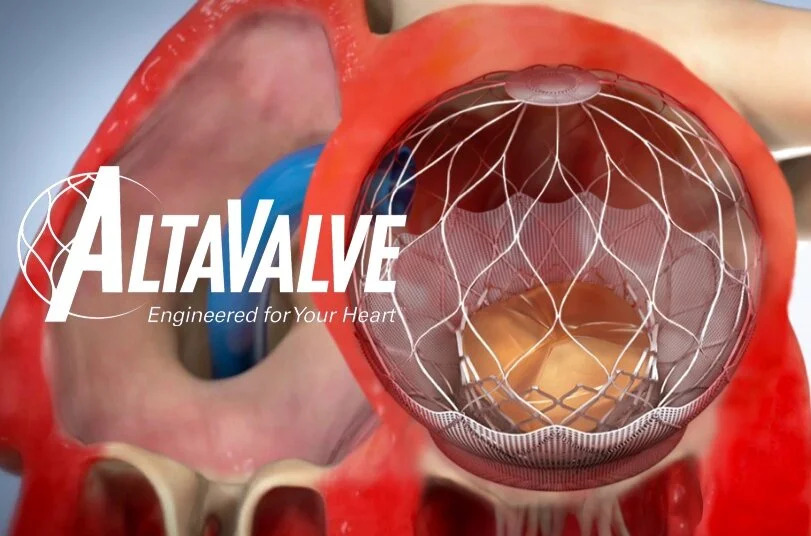
AltaValve System Shows Promise for Severe Mitral Regurgitation in 1-Year Results
www.medpagetoday.com
May 4, 2026, 7:09 a.m.
The AltaValve was designed to minimize LVOT obstruction and work around the anatomical limitations of current TMVR technologyopens in a new tab or window, namely the transapical Tendyneopens in a new tab or window and transseptal Sapien M3opens in a new tab or window systems currently on the market.Voudris noted that many TMVR systems rely on subvalvular fixation with rigid anchoring mechanisms. These increase the risk of LVOT obstruction and present anatomical limitations in people with complex mitral valve anatomy, large annuli, and mitral annular calcification (MAC), leading to infamously high screen failure ratesopens in a new tab or window associated with this technology.The AltaValve instead relies on atrial fixation. It features a bovine pericardium valve within a stent frame, topped with a stent cap that is attached to the delivery system. An annular ring is designed for no active attachment to native mitral valve or left ventricular structures, and the ring comes in three sizes.
Share on

Atrium-affixed TMVR system safe, improves 1-year outcomes
www.healio.com
May 4, 2026, 7:06 a.m.
A novel transcatheter mitral valve replacement system designed for left atrial fixation rather than subvalvular fixation improved mitral regurgitation and functional outcomes in patients at high surgical risk, a speaker reported.The system (AltaValve, 4C Medical Technologies) is designed to avoid left ventricular outflow tract (LVOT) obstruction and enable implantation in patients otherwise ineligible for transcatheter mitral valve replacement (TMVR) due to large valve annulus or mitral annular calcification, according to data presented at the Society for Cardiovascular Angiography & Interventions Scientific Sessions & Canadian Association of Interventional Cardiology/Association Canadienne de cardiologie d’intervention Summit.
Share on

HighLife TMVR system gains CE mark
cardiovascularnews.com
April 26, 2026, 6:54 p.m.
The initial commercial availability of the HighLife TMVR system across Europe provides access to a transfemoral mitral valve replacement option featuring a dual-component valve-in-ring design, capable of treating the broadest range of native mitral annulus sizes (30–53mm) of any CE mark–approved TMVR system.Clinical experience with the HighLife TMVR system includes patients treated across multiple countries, with longer-term follow-up data demonstrating durable outcomes, including sustained reduction of mitral regurgitation to mild or less; durable annular sealing enabled by a sub-annular ring fixation design, with no reported peri- or post-procedural paravalvular leakage PVL closure.
Share on

AltaValve early feasibility study reports out to one year
cardiovascularnews.com
April 26, 2026, 6:52 p.m.
Presenting the latest results during a late-breaking trials session at PCR London Valves (16–18 November, London, UK), Vlasis Ninios (Interbalkan Medical Center, Thessaloniki, Greece) reported that, in a population of patients including many who would have been ineligible to receive other mitral valve technologies, implanters achieved a high rate of technical success with the procedure, resulting in symptomatic improvements for patients.Left ventricular outflow tract (LVOT) obstruction is a common limitation of existing TMVR technologies leading to high rates of screen failure for patients screened for TMVR trials, Ninios highlighted in his presentation, ranking this alongside mitral annulus size and the presence of mitral annular calcification (MAC) as the exclusions that comprise the “Achilles’ heel” of existing TMVR technologies. AltaValve System’s atrial fixation TMVR device is designed to minimise the risk of LVOT obstruction and treat a broad population of mitral regurgitation (MR) patients as well as varied mitral annulus sizes.
Share on

New Study Shows Strong Outcomes for Transcatheter Tricuspid Valve Replacement
www.dicardiology.com
April 26, 2026, 6:35 p.m.
Tricuspid regurgitation occurs when the tricuspid valve does not close properly, allowing blood to flow backward in the heart. The condition is common in older adults and can lead to worsening heart failure symptoms, repeated hospitalization and increased risk of death.Historically, open-heart surgery was often the only treatment option for this condition, but many older patients were not candidates because of age or other medical problems. Today, transcatheter tricuspid valve replacement offers a less invasive alternative in which doctors replace the damaged valve using a catheter, or tube, threaded through a blood vessel.
Share on

First Implant of KingstronBio's ProStyle M® Transcatheter Mitral Valve System Successfully Completed in National Multicenter Confirmatory Study
www.prnewswire.com
April 8, 2026, 8:48 p.m.
Prior to this, ProStyle M® had undergone a two-year First-in-Man (FIM) study involving a total of 10 patients with a mean age of 71 years. The study results demonstrated excellent anchoring performance with no regurgitation observed in all cases. As the clinical study progresses, further evidence will be generated to verify the product's safety and efficacy.KingstronBio takes innovation as its core driving force and has long been deeply engaged in the heart valve field. The company owns a number of patented technologies at home and abroad, including the Micro-Ex™ anti calcification treatment technology and AirBo™ dry storage technology for heart valves.
Share on

La première implantation du système de valve mitrale transcathéter ProStyle M® de KingstronBio a été réalisée avec succès dans le cadre d'une étude nationale multicentrique de confirmation
www.prnewswire.com
April 8, 2026, 8:47 p.m.
Auparavant, ProStyle M® avait fait l'objet d'une étude First-in-Man (FIM) de deux ans portant sur un total de 10 patients dont l'âge moyen était de 71 ans. Les résultats de l'étude ont démontré une excellente performance d'ancrage, aucune régurgitation n'ayant été observée dans tous les cas. Au fur et à mesure que l'étude clinique progresse, des preuves supplémentaires seront produites pour vérifier l'innocuité et l'efficacité du produit.KingstronBio fait de l'innovation son principal moteur et est engagée depuis longtemps dans le domaine des valves cardiaques. L'entreprise possède un certain nombre de technologies brevetées dans le pays et à l'étranger, notamment la technologie de traitement anti-calcification Micro-Ex™ et la technologie de stockage à sec AirBo™ pour les valves cardiaques.
Share on

Transcatheter ViV Safe for Failed Bioprostheses
www.medscape.com
April 6, 2026, 10:31 a.m.
In a clinical trial comparing repeat mitral valve replacement surgery with a transcatheter valve-in-valve approach for dysfunctional bioprosthetic mitral valves, results linked the less invasive technique with a lower rate for death or stroke after 1 year. SURViV, is the first prospective clinical trial to compare the two procedures head-to-head. “Transcatheter mitral valve-in-valve has emerged as a less-invasive therapy for select high-risk patients,” he said. “Yet until now no trial has directly compared these two strategies.” The trial enrolled 150 patients with mitral bioprosthetic valve dysfunction between February 2020 and November 2023 and randomized them to redo surgery or transcatheter mitral valve-in-valve. The procedures were done at seven referral centers in Brazil. The primary composite endpoint of all-cause death and disabling stroke at 1 year was 20.8% for the surgery group and 5.3% for those who underwent valve-in-valve repair (P = .005), Siqueira said.
Share on