
Aspartame triggers genetic changes tied to glioblastoma severity
www.news-medical.net
July 14, 2025, 10:07 a.m.
Despite no visible tumor growth, new research finds that the artificial sweetener aspartame reshapes gut bacteria and upregulates cancer-linked genes in glioblastoma.
Share on

Vaccination Therapy Boosts Outcomes in Glioblastoma
bioengineer.org
July 7, 2025, 2:29 p.m.
Results indicated a statistically significant prolongation of progression-free survival among vaccinated patients, with a hazard ratio of 0.64 (p < 0.001). This suggests that vaccination therapies can reduce the risk of tumor progression by approximately 36% compared to controls. More intriguingly, a modest but highly significant improvement in overall survival was noted, with an HR of 1.09 (p < 0.00001). While the absolute survival benefit observed might appear modest, even incremental gains in glioblastoma are clinically meaningful, given the disease’s aggressive course and grim median survival times.
Share on
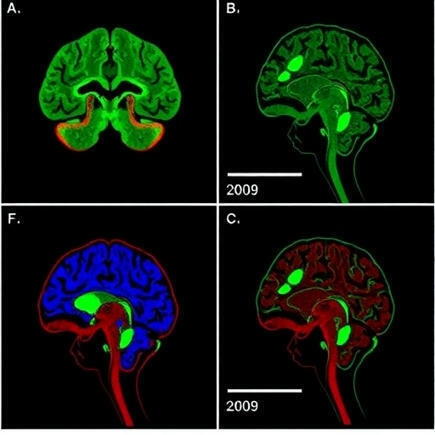
Gasdermin E Drives Pyroptosis Resistance in Glioblastoma
bioengineer.org
June 23, 2025, 8:47 a.m.
In a groundbreaking study poised to redefine our understanding of glioblastoma biology, researchers have uncovered a paradoxical role of Gasdermin E (GSDME) in this aggressive brain cancer. Traditionally recognized as a crucial mediator of pyroptosis—a highly inflammatory and lytic form of programmed cell death—GSDME has now been found to contribute to glioblastoma’s notorious resistance to pyroptosis, simultaneously promoting tumor progression. This unexpected duality challenges existing paradigms around cell death pathways in cancer and opens novel avenues for therapeutic intervention.
Share on

Glioblastoma patients live longer with triple treatment strategy
www.news-medical.net
June 14, 2025, 3:17 p.m.
The study finds that using Tumor Treating Fields therapy (TTFields), which delivers targeted waves of electric fields directly into tumors to stop their growth and signal the body's immune system to attack cancerous tumor cells, may extend survival among patients with glioblastoma, when combined with immunotherapy (pembrolizumab) and chemotherapy (temozolomide).
Share on
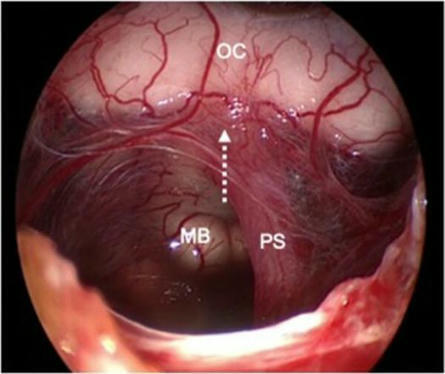
Is It Justified to Sacrifice the Pituitary Stalk During Craniopharyngioma Surgery? A Systematic Review and Meta-Analysis
www.neurosurgery-blog.com
June 14, 2025, 12:27 p.m.
This systematic review and meta-analysis examined the impact of pituitary stalk sacrifice during craniopharyngioma surgery. It found that sacrificing the stalk significantly increases the risk of postoperative endocrine dysfunction without reducing tumor recurrence or progression, highlighting the importance of preserving the stalk when possible.
Share on

Bivalent CAR-T Cells Via CSF Injection Show Activity in Recurrent Glioblastoma
www.medpagetoday.com
June 9, 2025, 6:26 a.m.
CAR-T activity is still modest in GBM, and there are multiple limitations to its use in this setting, said Elena Garralda, MD, PhD, of Vall d'Hebron Institute of Oncology in Barcelona, who served as ASCO session study discussant.
Share on
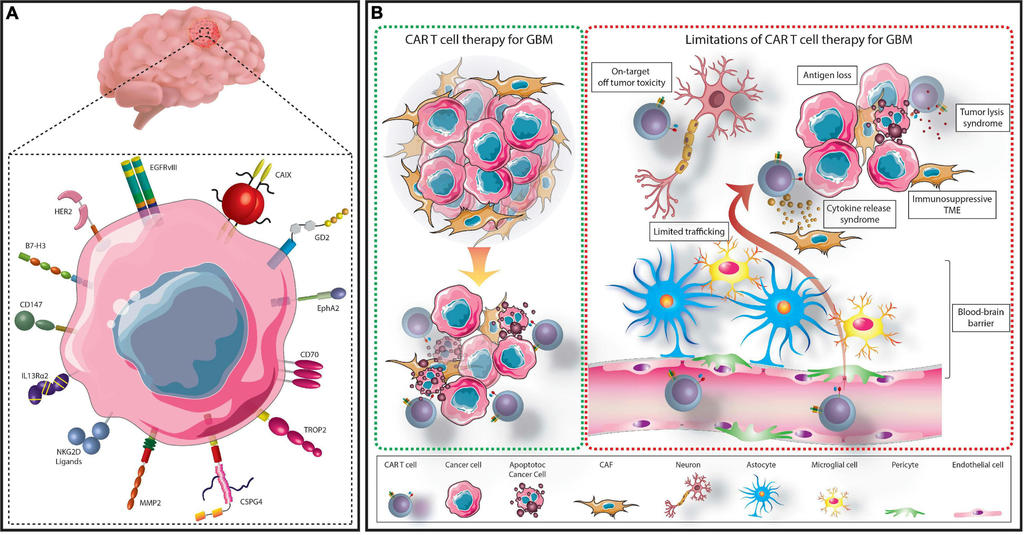
CAR T Cell-Based Immunotherapy for the Treatment of Glioblastoma
www.frontiersin.org
June 9, 2025, 6:26 a.m.
Characterization of GBM cells has contributed to identify several molecules as targets for immunotherapy-based treatments such as EGFR/EGFRvIII, IL13Rα2, B7-H3, and CSPG4. Cytotoxic T lymphocytes collected from a patient can be genetically modified to express a chimeric antigen receptor (CAR) specific for an identified tumor antigen (TA). These CAR T cells can then be re-administered to the patient to identify and eliminate cancer cells. The impressive clinical responses to TA-specific CAR T cell-based therapies in patients with hematological malignancies have generated a lot of interest in the application of this strategy with solid tumors including GBM.
Share on

New peptide therapy shows promise against glioblastoma recurrence
www.news-medical.net
June 3, 2025, 8:26 a.m.
In a study published in May in Cell Death and Disease, researchers identified a previously unknown trait of cancer cells that shows promise for therapeutic intervention. The group outlined the mechanism of action and effectiveness of the experimental drug known as JM2, revealing its potential as a peptide therapy to target cancer cells that can renew and regrow, even after chemotherapy and radiation.
Share on
Glioblastoma-Driven Astrocytes Suppress T Cells
bioengineer.org
May 26, 2025, 11:14 a.m.
In the relentless fight against glioblastoma, the most common and lethal form of primary brain cancer, new research is shedding light on a previously hidden collaborator within the tumor microenvironment—astrocytes. These star-shaped glial cells, traditionally known for their supportive roles in the central nervous system, have now been implicated in actively orchestrating immune evasion strategies that allow glioblastomas to thrive despite aggressive treatments. Groundbreaking work led by Faust Akl and colleagues unravels a complex molecular dialogue where tumor-derived signals reprogram astrocytes into suppressors of anti-tumor immunity, revealing promising therapeutic avenues that could reshape glioblastoma treatment paradigms.
Share on

Targeting astrocytes could boost immunotherapy effectiveness in glioblastoma
www.news-medical.net
May 26, 2025, 11:13 a.m.
Our study investigated the role of astrocytes, an abundant cell type in the brain, in regulating an immune response against glioblastoma (GBM)-a highly aggressive brain cancer. We found a subset of astrocytes that limits the immune response and can be targeted with therapeutics.
Share on
Gabapentinoids Improve Survival in Human Glioblastoma
bioengineer.org
May 19, 2025, 7:35 a.m.
In a groundbreaking study poised to shift the paradigm of glioblastoma treatment, researchers have unveiled compelling evidence that gabapentinoids, a class of drugs traditionally employed in neuropathic pain and seizure management, confer a significant survival advantage in human glioblastoma patients. This revelation arises from an extensive, multifaceted investigation that meticulously combines clinical data, molecular biology, and pharmacological insights, potentially opening new therapeutic avenues for one of the deadliest brain cancers known to medicine.
Share on

POSTN Splicing Epitopes Spark Hope in Glioblastoma Immunotherapy
bioengineer.org
May 19, 2025, 7:35 a.m.
In the relentless battle against gliomas, a notoriously aggressive and often deadly form of brain cancer, the quest for effective immunotherapy targets remains a paramount scientific challenge. Gliomas’ ability to evade immune detection has historically hindered the development of T-cell mediated therapies, largely due to the scarcity of identified tumor-specific antigens that effectively trigger immune responses. However, an innovative study is poised to change this narrative by unveiling a new reservoir of potential immunogenic targets derived from the aberrant transcriptomic landscape of glioma cells. This breakthrough work not only broadens our understanding of tumor antigenicity but also illuminates a promising avenue toward personalized immunotherapies.
Share on

Glioblastoma Vaccine Trial Moves Forward
neurosciencenews.com
May 12, 2025, 6:48 a.m.
The phase 2B SURVIVE trial testing SurVaxM, a cancer vaccine for glioblastoma, will continue as planned following an interim analysis showing sufficient promise to proceed. SurVaxM, combined with standard treatments like surgery, radiation, and chemotherapy, aims to extend survival and improve quality of life for patients with this aggressive brain cancer.The vaccine targets survivin, a protein that helps tumor cells evade death, and has shown a strong safety profile and encouraging survival rates in earlier studies. While specific results remain confidential due to trial regulations, the decision to continue marks a hopeful step in the development of more effective glioblastoma therapies.
Share on

Needle Biopsies Enable Multimodal Data in Glioblastoma
bioengineer.org
May 5, 2025, 6:16 a.m.
In a groundbreaking advancement that promises to reshape the landscape of glioblastoma research and treatment, a team of researchers led by Yu, Basu, Baquer, and their colleagues have unveiled a novel investigative approach utilizing needle core biopsies to enable comprehensive multimodal deep-data generation.
Share on

A zinc transporter drives glioblastoma progression via extracellular vesicles-reprogrammed microglial plasticity
www.pnas.org
May 5, 2025, 6:15 a.m.
Glioblastoma (GBM) remains a formidable clinical challenge, underscoring the urgent need for new therapeutic strategies. This study identifies ZIP4, a zinc transporter, as a pivotal regulator of microglial plasticity and immune landscape remodeling within the tumor microenvironment. By revealing ZIP4’s role in driving tumor progression and immune modulation, this work positions ZIP4 as a promising therapeutic target for combating this aggressive and debilitating cancer.
Share on
GSK Grabs Rights to Technology That Gets Drugs Across the Blood-Brain Barrier
medcitynews.com
April 19, 2025, 4:15 p.m.
GSK is licensing an ABL Bio technology that yields bispecific antibodies engineered to leverage a certain transmembrane receptor to cross the blood-brain barrier to treat neurodegeneration. GSK is already partnered with Alector, which has monoclonal antibodies in clinical development for Alzheimer’s disease.
Share on
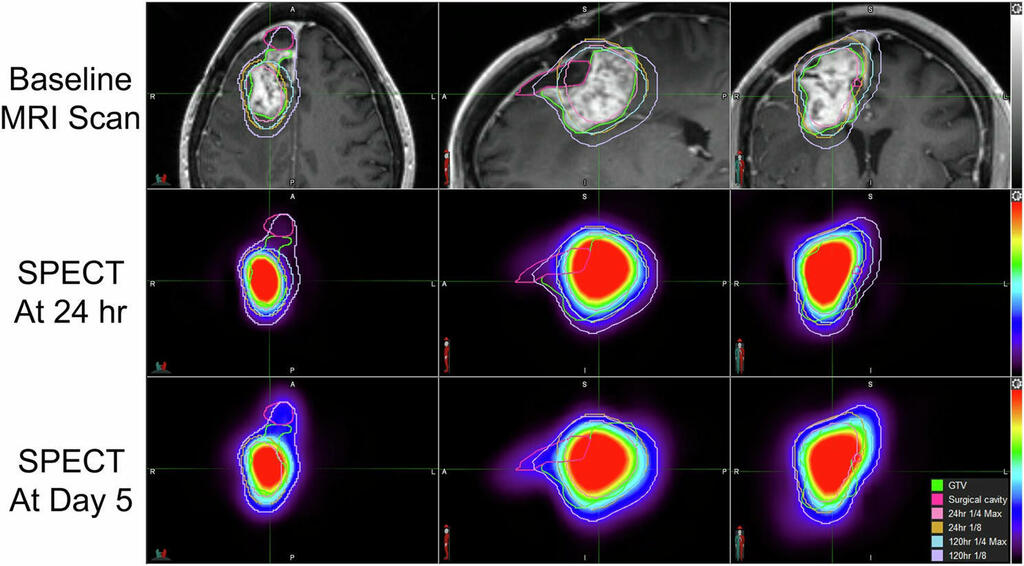
Nanoparticles extend glioblastoma survival in phase one trial
medicalxpress.com
April 7, 2025, 7:59 a.m.
In a Phase I clinical trial reported in Nature Communications, two researchers from UT Southwestern and their colleagues showed this strategy was safe and effective. The team worked with 21 patients at medical centers, including UTSW, who had recurrent glioblastoma. They were divided into six groups, each of which received a different dose of radiation-emitting nanoparticles through CED. Patients who received the highest doses had tolerable side effects and lived an average of 17 more months after treatment, significantly longer than expected for patients with recurrent glioblastoma. The authors suggest this strategy shows promise for improving treatments for these patients.
Share on

Epigenetic reprogramming of glioblastoma to overcome chemotherapy resistance
www.nature.com
April 7, 2025, 7:58 a.m.
As recently reported in Neuro-Oncology, a team led by John Liu at the University of California, San Francisco Brain Tumor Center has developed a CRISPR–Cas9-based epigenetic editing approach that sensitizes glioblastoma cells to standard chemotherapy drugs. This technology could improve the treatment of tumours that have developed resistance to these drugs.
Share on
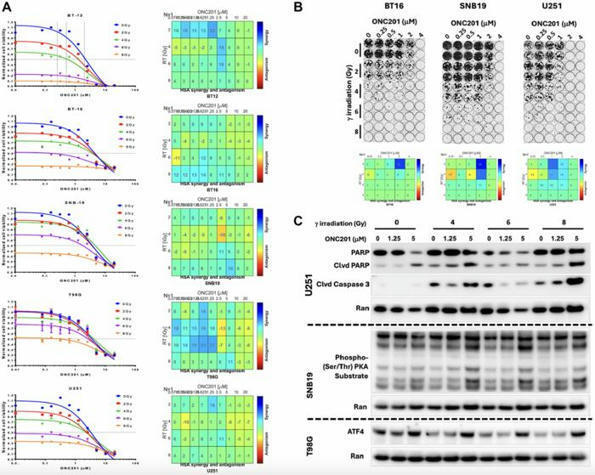
Triple Therapy Demonstrates Potential to Inhibit Glioblastoma Progression
bioengineer.org
April 7, 2025, 7:58 a.m.
Researchers are continuously on the lookout for innovative treatment strategies for glioblastoma, a notoriously aggressive brain cancer. Recently, an exciting preclinical study published in the esteemed journal Oncotarget explored the combination of imipridones—specifically, ONC201 and its analog ONC206—with traditional therapies like radiation (RT) and temozolomide (TMZ). The research team, led by Brown University’s Lanlan Zhou under the guidance of Wafik S. El-Deiry, focuses on a revolutionary treatment regimen termed IRT—imipridones, radiation, and temozolomide. This therapy has demonstrated a potential breakthrough in reducing tumor burden and prolonging survival in an orthotopic IDH-WT glioblastoma mouse model.
Share on
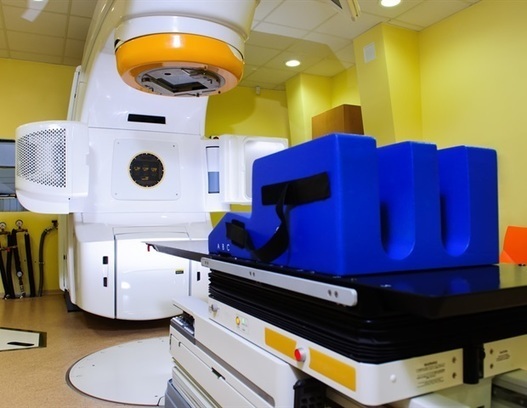
Triple therapy reduces tumor burden and improves survival in glioblastoma models
www.news-medical.net
April 7, 2025, 7:57 a.m.
A new research paper was published in Oncotarget, Volume 16, on March 27, 2025, titled "Imipridones ONC201/ONC206 + RT/TMZ triple (IRT) therapy reduces intracranial tumor burden, prolongs survival in orthotopic IDH-WT GBM mouse model, and suppresses MGMT."
Share on