
But do we need high bandwidth? Applications and scaling challenges of invasive brain–computer interfaces
iopscience.iop.org
June 8, 2026, 9:30 a.m.
For today’s clinical goals (reliable communication and functional motor restoration), moderate bandwidth already suffices when coupled with model-based priors, structured output spaces, and shared-control architectures; next-horizon goals, e.g. unconstrained natural speech, embodied dexterity, and cognitive restoration, however, require abundant sampling but remain constrained by biological, technical, and ethical hurdles, with the engineering trilemma of bandwidth, power, and latency as the primary bottleneck for fully implantable systems. Solving this requires a shift towards low-power on-implant processing to handle increasing neural datastreams.
Share on
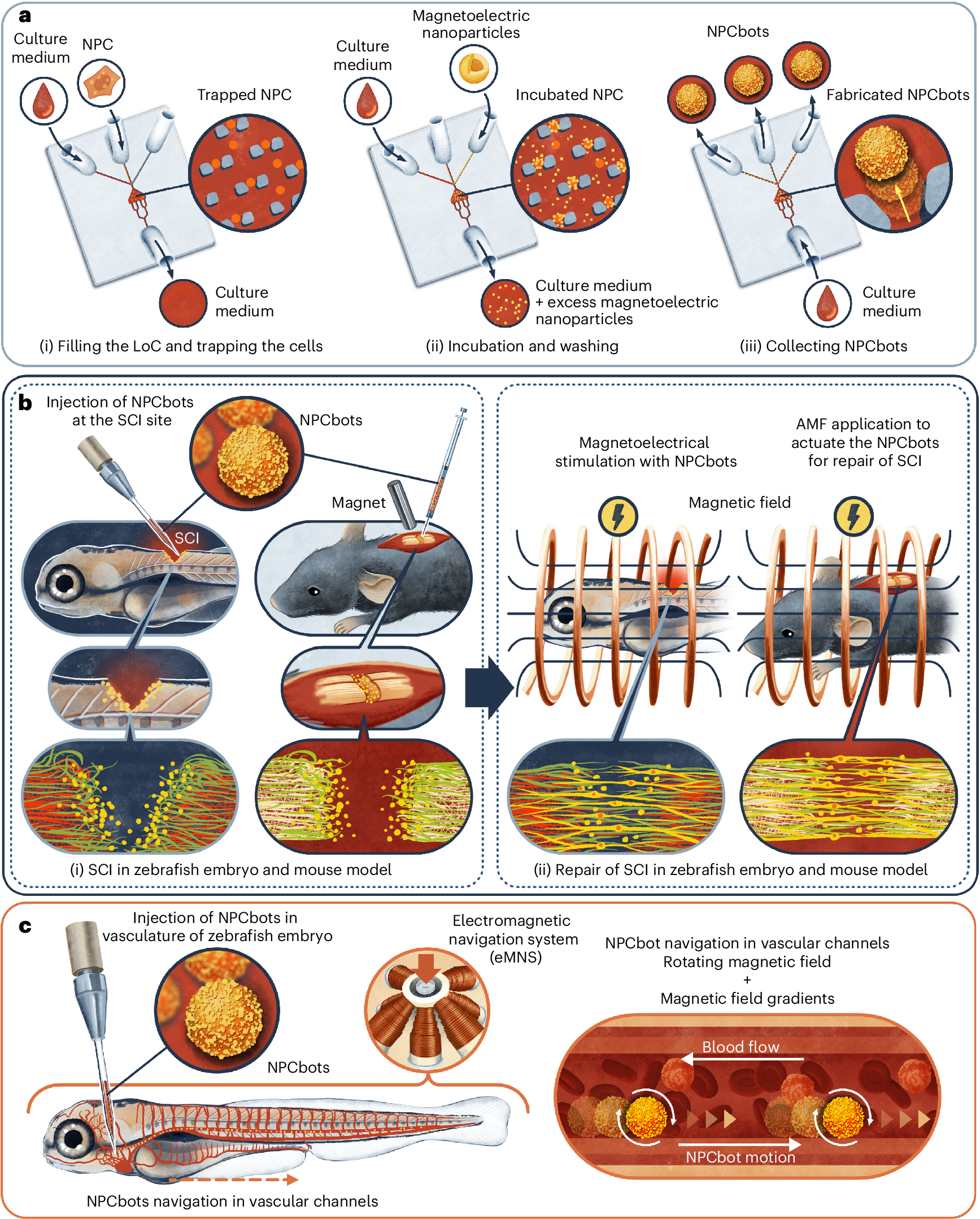
Magnetic microrobots deliver stem cells to heal spinal cord injuries
www.news-medical.net
June 8, 2026, 6:12 a.m.
Researchers at ETH Zurich and the University of Zurich have developed an innovative treatment for spinal cord injuries using magnetic microrobots combined with stem cells. The biohybrid microrobots integrate neural progenitor cells derived from induced pluripotent stem cells with magnetoelectric nanoparticles, enabling precise navigation to injury sites through magnetic guidance. Once positioned, the nanoparticles convert magnetic signals into electrical stimulation, promoting nerve cell regeneration and tissue repair. Animal experiments demonstrated significantly improved mobility outcomes. This approach overcomes limitations of existing therapies by eliminating the need for implanted electrodes while ensuring better cell survival and integration. The research, published in Nature Materials, represents a promising advancement in regenerative medicine for treating spinal cord injuries.
Share on
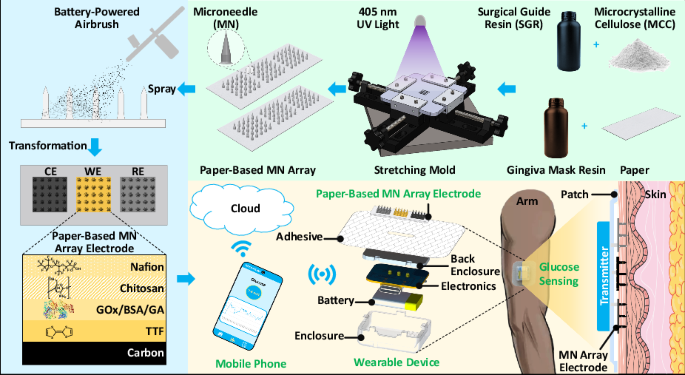
A wearable paper-based SGR/MCC microneedle array sensor for ...
www.nature.com
June 8, 2026, 6:12 a.m.
Researchers have developed an innovative wearable paper-based microneedle array sensor for continuous glucose monitoring in diabetes patients. The device features solid microneedles fabricated from a biocompatible composite of surgical guide resin and microcrystalline cellulose, mounted on a flexible paper substrate designed for comfort. The microneedles are converted into conductive biosensing electrodes through graphene coating and functional modifiers, enabling electrochemical glucose detection in dermal interstitial fluid. The integrated system combines disposable microneedle arrays with reusable electronics and mobile application support. Testing demonstrates excellent analytical performance with high sensitivity and selectivity across multiple models, including ex vivo porcine plasma and in vivo mice studies, presenting a practical advancement toward practical continuous glucose monitoring solutions.
Share on
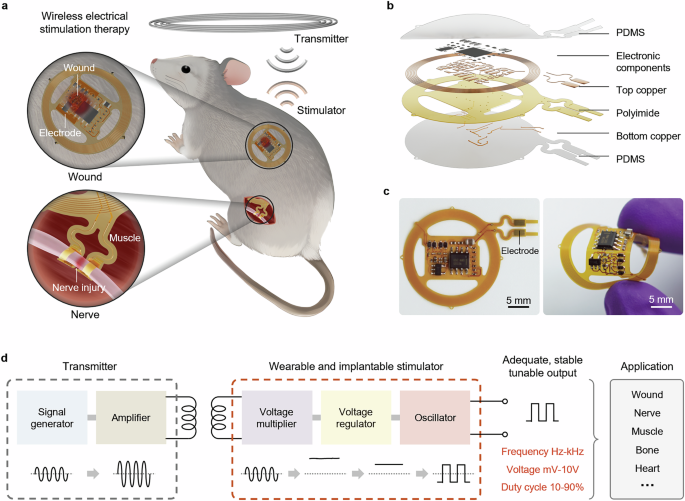
A wireless, position-insensitive electrical stimulation platform with ...
www.nature.com
June 8, 2026, 6:11 a.m.
Researchers have developed an innovative wireless, battery-free electrical stimulation platform that addresses significant limitations in existing therapeutic devices. The system utilizes inductive coupling and advanced radio-frequency circuit design to deliver high-voltage stimulation up to 15 volts while maintaining remarkable tolerance to physical misalignment, functioning effectively even with lateral shifts of 18 millimeters, vertical displacement of 15 millimeters, and angular variations up to 40 degrees. Featuring highly configurable parameters including adjustable amplitude, frequency ranging from hertz to kilohertz, and variable duty cycles, this versatile platform accommodates diverse clinical applications. Demonstrated applications include epidermal wound healing and peripheral nerve regeneration, with in vitro studies confirming enhanced cellular proliferation, migration, and neurite extension, positioning this technology as a promising minimally invasive approach for tissue repair and treatment of various pathological conditions.
Share on

Announces IDE Submission for U.S. Clinical Study of its Novel Implantable Continuous Blood Glucose Monitoring Technology
glucotrack.com
June 1, 2026, 1:36 p.m.
Glucotrack’s Continuous Blood Glucose Monitor (CBGM) is a long-term, implantable system that continually measures blood glucose levels with a sensor longevity of 3 years, no on-body wearable component and with minimal calibration. The Glucotrack CBGM is an Investigational Device and is limited by federal (or United States) law to investigational use.

MindVoice: Reconstructing Intelligible Speech from Non-invasive ...
arxiv.org
June 1, 2026, 1:33 p.m.
MindVoice presents a groundbreaking neuro-to-speech reconstruction framework that successfully transforms non-invasive neural signals into intelligible speech. Addressing the challenge of inherent noise and spatial blur in EEG and MEG recordings, the system employs a dual-pathway approach: one channel recovers high-level semantic content while the other estimates acoustic attributes. By leveraging pretrained models as compensatory priors, MindVoice bridges the information gap between incomplete neural data and natural speech synthesis. The framework integrates advanced speech generation models with in-context voice cloning to produce coherent, natural utterances. Extensive experimental validation demonstrates substantial performance improvements over existing methods, positioning this work as a significant advancement for auditory neuroscience research and practical non-invasive speech brain-computer interface applications.
Share on
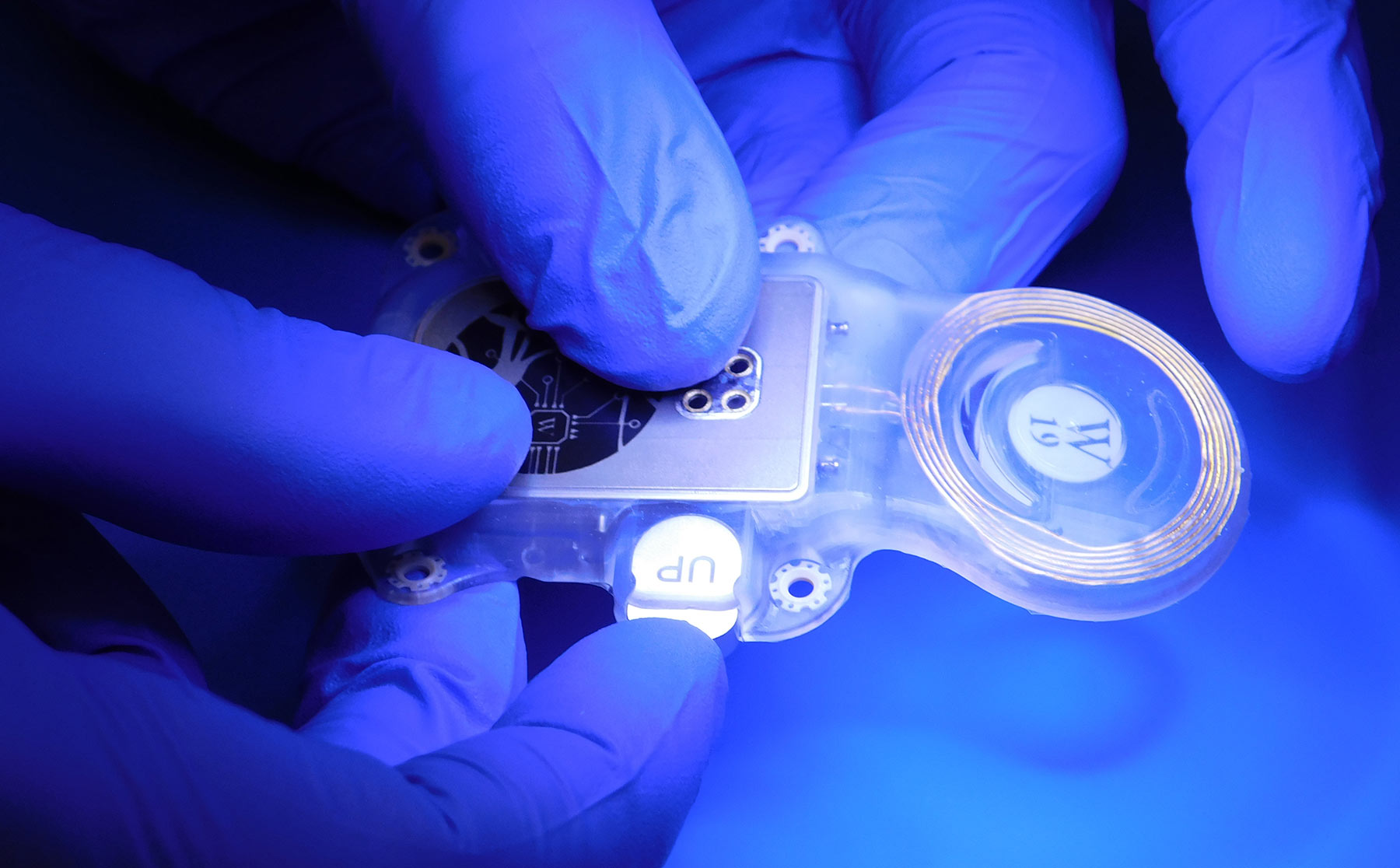
ABILITY Neurotech Receives IMDD Approval to Start Clinical Trial ...
finance.yahoo.com
June 1, 2026, 1:31 p.m.
ABILITY Neurotech has secured Investigational Medical Device Dossier approval from the Medical Research Ethics Committee in the Netherlands to commence its first chronic implantation clinical trial of its fully implantable wireless optical-link ECoG brain-computer interface in amyotrophic lateral sclerosis patients. This significant milestone represents a transition from intraoperative testing to long-term human clinical investigation, validating the company's comprehensive preclinical data and compliance with European Medical Device Regulation requirements. The study will be conducted at UMC Utrecht in collaboration with the INTRECOM consortium, including the Technical University of Graz and CorTec, focusing on evaluating the BCI's performance as a home-use system to restore communication and speech capabilities in ALS patients.
Share on
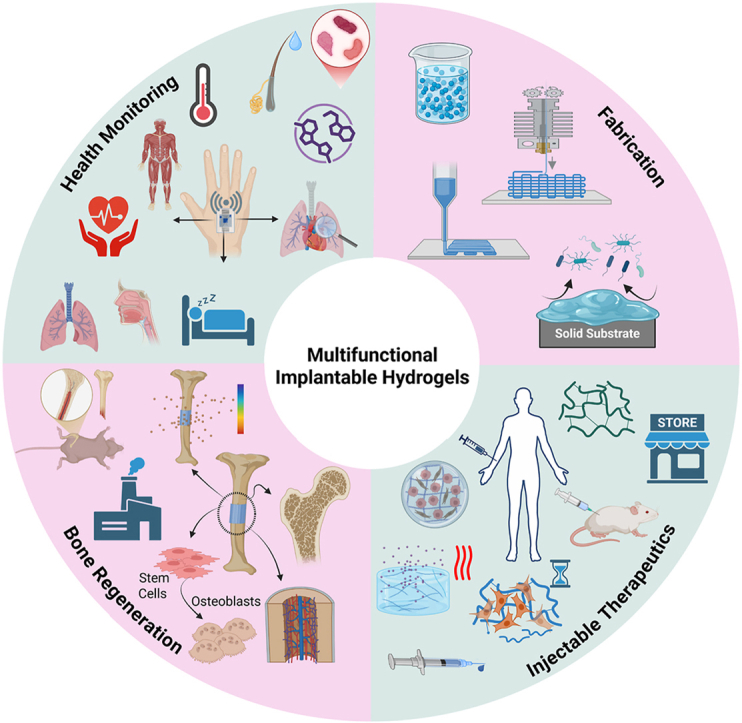
Multifunctional implantable hydrogels: Smart platforms at the ...
pmc.ncbi.nlm.nih.gov
June 1, 2026, 9:07 a.m.
Multifunctional implantable hydrogels represent a significant advancement in biomedical technology, serving as intelligent platforms that integrate multiple therapeutic and diagnostic capabilities. These smart biomaterials demonstrate considerable promise in revolutionizing medical treatment approaches by combining biocompatibility with responsive functionality. Authored by leading researchers from international institutions including the University of Sydney and Alexandria University, this comprehensive review examines the innovative applications and technological developments of these hydrogel systems. The article explores how these platforms address critical clinical challenges through their unique ability to deliver drugs, monitor physiological parameters, and facilitate tissue regeneration simultaneously. As the field advances toward personalized medicine, multifunctional implantable hydrogels are positioned to transform patient care outcomes across diverse medical specialties.
Share on
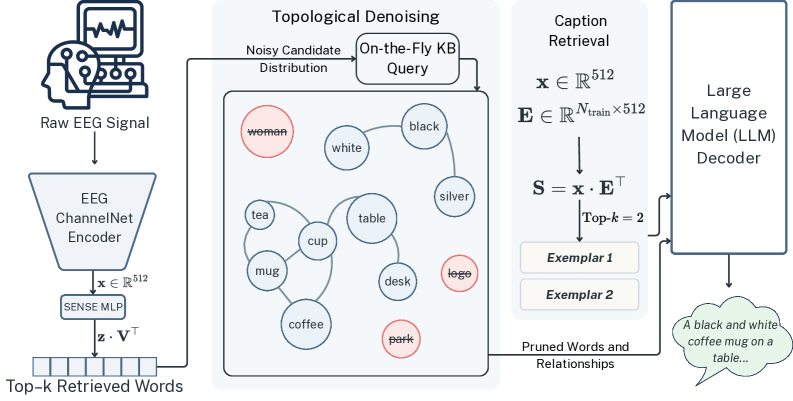
SYNAPSE: Neuro-Symbolic Visual Thought-to-Text Decoding ...
arxiv.org
June 1, 2026, 9:06 a.m.
SYNAPSE is a neuro-symbolic framework that decodes visual thoughts from EEG neural activity into coherent text descriptions. Addressing vulnerabilities in existing systems prone to biological noise and semantic instability, SYNAPSE employs inference-time symbolic regularization using commonsense graphs and latent exemplars to purify semantic candidates without requiring computationally expensive language model fine-tuning. The lightweight approach demonstrates consistent performance improvements across multiple EEG decoding benchmarks and frozen language model backends, matching more resource-intensive alternatives while maintaining biometric privacy through localized EEG processing.
Share on

FDA-Approved Vivistim: A New Pathway in Stroke Recovery
medicaldevicesandpharma.com
June 1, 2026, 9:06 a.m.
Mobia Medical's Vivistim® Paired VNS™ System represents a significant advancement in stroke rehabilitation, recently securing FDA approval through the Premarket Approval pathway with Breakthrough Device designation. This implantable vagus nerve stimulation device combines neurostimulation therapy with structured rehabilitation to enhance motor recovery in chronic ischemic stroke patients experiencing moderate to severe upper-extremity deficits. Unlike conventional rehabilitation-only approaches, Vivistim offers a clinically validated, innovative treatment option supported by robust evidence. The company's recent public offering, raising approximately $150 million, signals strong confidence in the technology's commercial potential and reflects growing market recognition of its therapeutic value in neurorehabilitation.
Share on
Editorial: Non invasive BCI for communication - Frontiers
www.frontiersin.org
May 8, 2026, 7:46 p.m.
Non-invasive brain-computer interfaces represent a transformative advancement in restoring communication for individuals with severe motor disabilities. Unlike invasive surgical alternatives, these systems offer enhanced accessibility and safety while supporting broader deployment potential. Communication-focused BCIs must effectively convert brain signals into reliable outputs such as spelling and selection without requiring muscle activity. Electroencephalography dominates current applications due to its portability and superior temporal resolution, though alternative modalities are being explored to enhance system robustness. Despite significant progress in BCI technology, challenges persist in translating these systems beyond controlled laboratory environments, highlighting the ongoing need for further development and innovation in this critical field.
Share on
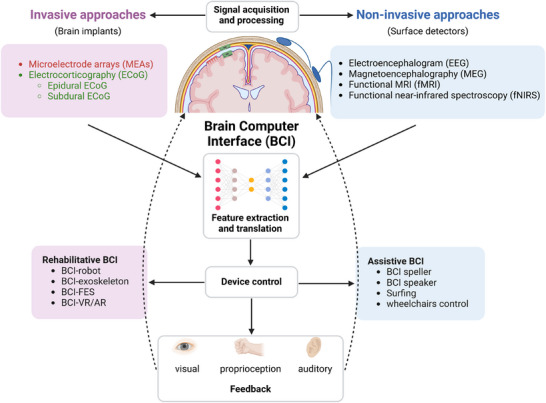
Emerging Neural Recording and Neurostimulation Technologies ...
pmc.ncbi.nlm.nih.gov
May 8, 2026, 7:46 p.m.
Brain-computer interface technologies represent a transformative frontier in treating neuropsychiatric disorders through advanced neural recording and neurostimulation capabilities. These emerging technologies enable direct communication between the brain and external devices, facilitating precise monitoring of neural activity and targeted therapeutic interventions. By leveraging sophisticated recording techniques and stimulation protocols, researchers can address conditions including depression, anxiety, and neurological disorders with unprecedented precision. This approach offers significant advantages over conventional treatment modalities, particularly for patients with treatment-resistant conditions. As the field advances, brain-computer interface applications demonstrate considerable promise in revolutionizing neuropsychiatric care, improving patient outcomes, and expanding therapeutic options for previously intractable neurological and psychiatric conditions.
Share on
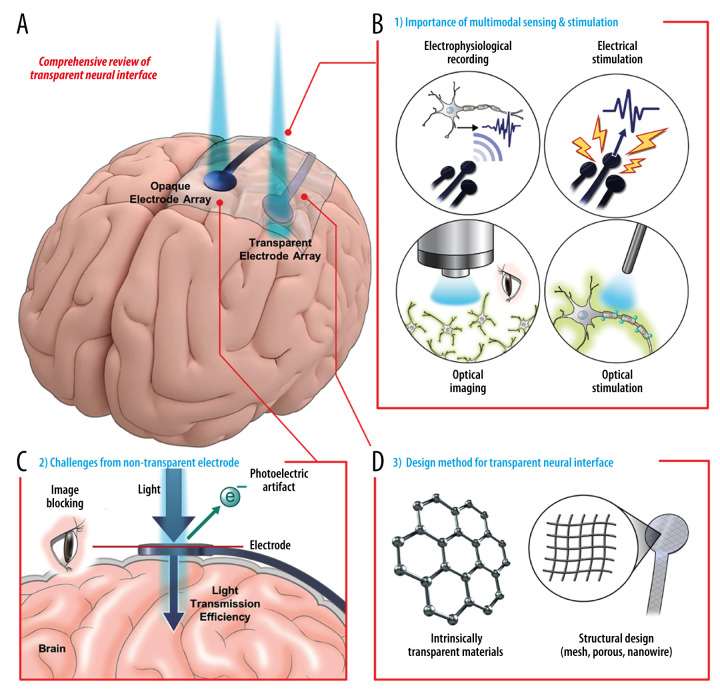
Review of Recent Advances in Implantable Brain-Computer ... - PMC
pmc.ncbi.nlm.nih.gov
May 8, 2026, 7:46 p.m.
Recent advances in implantable brain-computer interfaces represent a significant breakthrough in neurorehabilitation, offering promising solutions for patients suffering from paralysis. These sophisticated technologies enable direct communication between the brain and external devices, effectively bypassing damaged neural pathways to restore motor function. The review examines cutting-edge developments in implantable interface design, signal processing algorithms, and clinical applications, highlighting the transformative potential of this emerging field. As researchers continue to refine these systems, implantable brain-computer interfaces demonstrate increasing efficacy in restoring voluntary movement and improving quality of life for individuals with severe motor impairments, marking a pivotal advancement in regenerative neurology.
Share on

China Clears First Brain-Computer Implant for Commercial Use
www.sixthtone.com
May 8, 2026, 7:46 p.m.
China's National Medical Products Administration has approved the NEO system, developed by Shanghai-based Neuracle Medical Technology, marking the world's first invasive brain-computer interface cleared for commercial clinical use. The device combines an implanted BCI with an EEG electrode kit and pneumatic robotic glove to restore limited hand movement in quadriplegic patients with cervical spinal cord injuries. Clinical trials involving 36 participants demonstrated improved hand grasping ability across all subjects, with some showing signs of neural remodeling suggesting potential recovery of additional neurological function. The system operates by detecting neural signals associated with movement intent and wirelessly transmitting them to the robotic glove, enabling patients to grasp and release objects. Shanghai has announced three new research centers to accelerate the technology's further development.
Share on

CorTec receives FDA breakthrough designation for brain-computer interface in stroke rehab
roboticsandautomationnews.com
April 18, 2026, 8:17 a.m.
CorTec’s Brain Interchange system combines neural signal recording with adaptive stimulation in a closed-loop architecture. Unlike BCIs focused solely on enabling communication through external devices, the system is designed to both interpret brain signals and deliver therapeutic stimulation aimed at restoring motor function.The platform is currently being evaluated in an FDA-approved investigational device exemption (IDE) study at the University of Washington in Seattle. According to the company, this represents the first clinical investigation of a fully implantable, wireless BCI system for stroke rehabilitation in humans.
Share on

Did Neuralink make the wrong bet?
www.theverge.com
April 18, 2026, 8:16 a.m.
Elon Musk promised Neuralink would bring superhuman abilities and minds merged with AI. Then he fueled a runaway hype train for his brain implant technology, which ended up with a grisly record for implants in monkeys and some success with human subjects. But for all of the hype, he’s still further away than Mars from his goal. And that’s because his relentless ambition is once again hitting the wall of scientific reality.
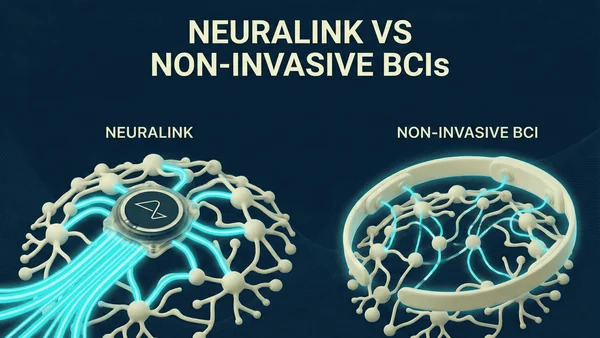
Neuralink a-t-il fait le mauvais pari ?
france-jeunes.net
April 18, 2026, 8:14 a.m.
L'interface cerveau-ordinateur (BCI) ne relève plus de la science-fiction, mais la course effrénée lancée par Elon Musk avec Neuralink a-t-elle sacrifié la prudence médicale sur l'autel du spectacle technologique ? Alors que les annonces médiatiques fascinent le grand public, la réalité clinique des implants révèle des défis structurels majeurs qui remettent en question la viabilité de l'approche « tout ou rien ». Neuralink a-t-il fait le mauvais pari en misant tout sur une technologie invasive et complexe au détriment de la sécurité et de la scalabilité ? Nous analysons ici les implications techniques, biologiques et éthiques de cette stratégie audacieuse à la lumière des récents résultats cliniques.
Share on

The BCI User Experience: Living With Brain Implants
spectrum.ieee.org
April 18, 2026, 8:11 a.m.
In 1985, Imbrie had woken up in the hospital after a car accident with a broken neck and a doctor telling him he’d never use his hands or legs again. His response was an expletive, he says—and a decision. “I’m not going to allow someone to tell me what I can and can’t do.” With the determination of a head-strong 22-year-old, Imbrie gradually regained the ability to walk and some limited arm movement. Aware of how unusual his recovery was, the Illinois-native wanted to help others in similar situations and began looking for research projects related to spinal cord injuries. For decades, though, he wasn’t the right fit, until in 2020 he was finally accepted into a University of Chicago trial.
Share on

Scientists Develop Two-Way Brain Interface with Wearable Robotic Legs to Restore Walking and Sensation After Paralysis
bioengineer.org
April 18, 2026, 8:10 a.m.
In a groundbreaking convergence of neuroscience and robotics, researchers from the Keck School of Medicine of USC, the University of California, Irvine (UCI), and the California Institute of Technology (Caltech) have propelled the ambitious quest to restore walking and sensation in patients with paraplegia forward. Their innovative work harnesses the power of a fully implantable brain-computer interface (BCI) integrated with a wearable robotic exoskeleton, marking a significant leap towards reestablishing natural, bidirectional communication between the brain and limbs once paralyzed.
Share on

Startup Develops Brain-Reading Wearable to Convert Thoughts into Text
killbait.com
April 18, 2026, 8:09 a.m.
California-based startup Sabi is developing a noninvasive brain-computer interface (BCI) that converts a person’s internal speech into text displayed on a computer.Unlike companies such as Neuralink that focus on surgically implanted devices, Sabi aims to make this technology accessible to the general public through wearable devices like a beanie and a baseball cap.The device relies on electroencephalography (EEG) to detect brain activity, and Sabi plans to use 70,000 to 100,000 miniature sensors to improve signal accuracy. The initial typing speed is projected at around 30 words per minute, with improvements expected as users become accustomed to the device.To handle the variability in individual thought patterns, Sabi is creating a large-scale AI model, called a brain foundation model, trained on extensive neural data from many volunteers. Consumer usability is a major focus, with an emphasis on comfort, ease of use, and out-of-the-box functionality.
Share on